 
Thirdly, the intermediate and ultimate hydrolytic breakdown products of PPEs are innocuous, conferring PPEs excellent toxicity and biocompatibility profiles. Secondly, the flexible phosphoester bond in the backbone also provides the plasticizing effect to lower the glass transition temperature and improve solvent solubility and processibility of polymers with otherwise unfavorable physical and colligative properties. The phosphorus-containing backbone confers versatility and tunability in PPE-based material design as the physicochemical and degradation properties can be adjusted by changing the backbone and the side-chain structures. These are tabulated on the basis of their applications in Table 1 (biodegradation (1-1 to 1-3), drug and gene carriers (2-1 to 2-5), and thermoresponsive PPEs (3-1 to 3-6)). Starting from the parent polyphosphite, the P–H bonds can be readily converted for conjugation of different chemical structures, including charged groups through a phosphate (P–O) or a phosphoramide (P–N) bond as side chains. 
The pentavalency of phosphorus in the backbone of PPEs makes it possible to conjugate functional groups.  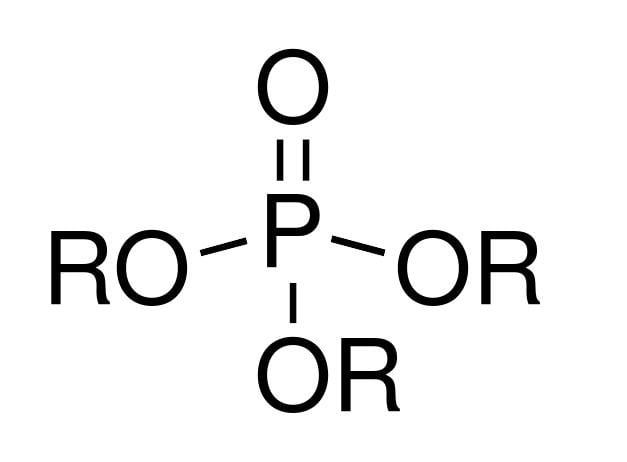
51–53 Here, we highlight the characteristics of PPEs that will repeatedly manifest in various biomedical applications discussed in this chapter. 50 Much of the research into the basic science of these polymers has been pioneered by Penczek and his colleagues. PPEs represent a class of biodegradable polymers having a phosphoester linkage in the backbone, which would include polyphosphates, polyphosphonates, and polyphosphites. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
